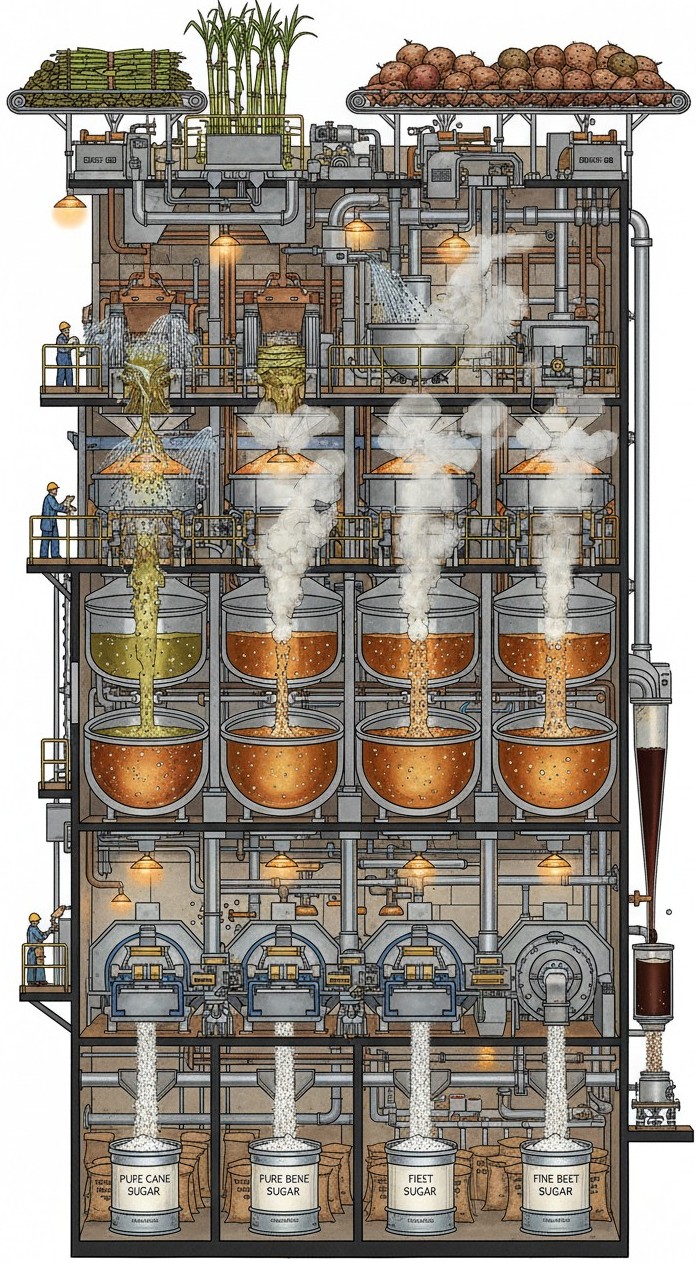
Professional intake for buyers, distributors, institutions, procurement teams, healthcare groups, and export partners seeking structured pricing review for SAFE SUGAR™ bulk supply.
This page is built to collect structured commercial details before formal quotation review. It supports more serious buyer conversations and reduces low-quality or incomplete inquiries.
SAFE SUGAR™ is not positioned or quoted like commodity sugar. Serious submissions with clear quantity, destination, packaging, and use-case detail receive stronger review and better follow-up.
For qualified distributors, importers, wholesalers, and market-access partners seeking structured supply review.
For hospitals, healthcare groups, and government or institutional buyers evaluating supply pathways.
For commercial teams planning recurring demand, formulation integration, or container-scale movement.
For buyers who can provide clear destination, port, timing, and commercial assumptions for export review.
To maintain positioning, quality, and supply discipline, SAFE SUGAR™ quotations are issued only for serious institutional and commercial inquiries.
The standard minimum order quantity for SAFE SUGAR™ is 5 metric tons. Smaller retail, trial, or low-volume resale requests are not the focus of this supply program.
Quotations are structured based on application, order volume, destination country, packaging format, shipping terms, and deployment scope. SAFE SUGAR™ is not quoted as a commodity sugar product.
Shipping terms may be structured on an EXW, FOB, or CIF basis, depending on the order, destination, and commercial arrangement. Final shipping structure is confirmed during quotation review.
Standard fulfillment timelines are generally in the range of 8 to 12 weeks, depending on current production scheduling, order size, destination, packaging requirements, documentation, and shipping conditions.
Qualified orders are generally handled on a deposit-based production structure. Standard terms are typically 50% upfront deposit to secure production, with the remaining balance due according to the final commercial agreement before release or shipment.
Qualified submissions can be reviewed for routing, fit, supply assumptions, seriousness of inquiry, and quotation response. Submission does not automatically guarantee supply, exclusivity, or production allocation.
No. This page is built for commercial, distributor, industrial, institutional, and export use cases. It is not intended as a general consumer contact form.
Complete the fields below with serious commercial detail. The better the information, the stronger the quotation review and follow-up quality.
The buyer submits company, destination, quantity, packaging, timing, and commercial detail.
The inquiry is reviewed for seriousness, route fit, supply assumptions, and deployment alignment.
Qualified submissions can receive structured follow-up, pricing review, and shipping-term discussion.
Approved orders move into deposit-based production planning and coordinated commercial execution.
For direct business communication related to SAFE SUGAR™ quotation review, use the official Lathell Nutraceuticals LLC contact details below.



